Usually, when we refer to density we mean a mass density which would be mass per unit volume. This is usually expressed by the equation
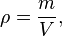 where the Greek letter ρ stands for density, m is for mass, and V for volume. Some texts will use the letter D for density.
where the Greek letter ρ stands for density, m is for mass, and V for volume. Some texts will use the letter D for density.
In the metric system we might use units of grams per milliliter, grams per cubic centimeter or kilograms per cubic meter. It is worth noting that the c.c or cubic centimeter is equal to one milliliter. You often hear the term c.c. on bad television shows about hospital emergency rooms.
However, we could also have a charge density, current density, energy density among many others!
The density in a solid object does depends not only on the mass of the individual atoms, but on how those atoms are arranged. What kind of crystal lattice creates spaces between the atoms, or is the solid an amorphous blob?
It is worth noting that the density of water happens to be 1 g/ml or 1000 kg/cu.m This is largely due to the historical context and the role water played in defining the gram.
 Most metals have a density about 10 times greater than water. This table of densities is in units of g/ml
Most metals have a density about 10 times greater than water. This table of densities is in units of g/ml
| Solids | ||
| Magnesium | 1.7 | |
| Aluminum | 2.7 | |
| Copper | 8.3-9.0 | |
| Gold | 19.3 | |
| Iron | 7.8 | |
| Lead | 11.3 | |
| Platinum | 21.4 | |
| Uranium | 18.7 | |
| Osmium | 22.5 | |
| Ice at 0 C | 0.92 | |
In your ancient history class you probably will read the Eureka story of Archimedes. As the story goes, a dishonest goldsmith mixed some other metals into a crown made for Hiero of Syracuse. The King asked Archimedes to determine if it was true gold. Archimedes was able to measure the weight, but not the volume as the crown had an irregular shape. While taking a bath, Archimedes stumbled upon the solution to the problem and ran through the streets of Syracuse naked. In truth, Archimedes never wrote about this and the story can be attributed to Vitruvious. You should read the passages by Vitrivious which are in paragraphs 9-12. Of course, the entire story is somewhat suspect as elaborated by this article in Scientific American.

I’ll end this with a couple thoughts. Why is it impossible to sink in the dead sea?
And what is happening in this picture? Why don’t the colours mix in this cylinder?
Reminds me a bit of a Tequila Sunrise.
One last thing to share is a great YouTube video from a Museum up in Canada.

